Piperazine ring toxicity in three novel anti-breast cancer drugs: an in silico and in vitro metabolic bioactivation approach using olaparib as a case study | SpringerLink

Science of Synthesis Knowledge Updates – Review on Piperazine Synthesis - Thieme Chemistry - Georg Thieme Verlag KG
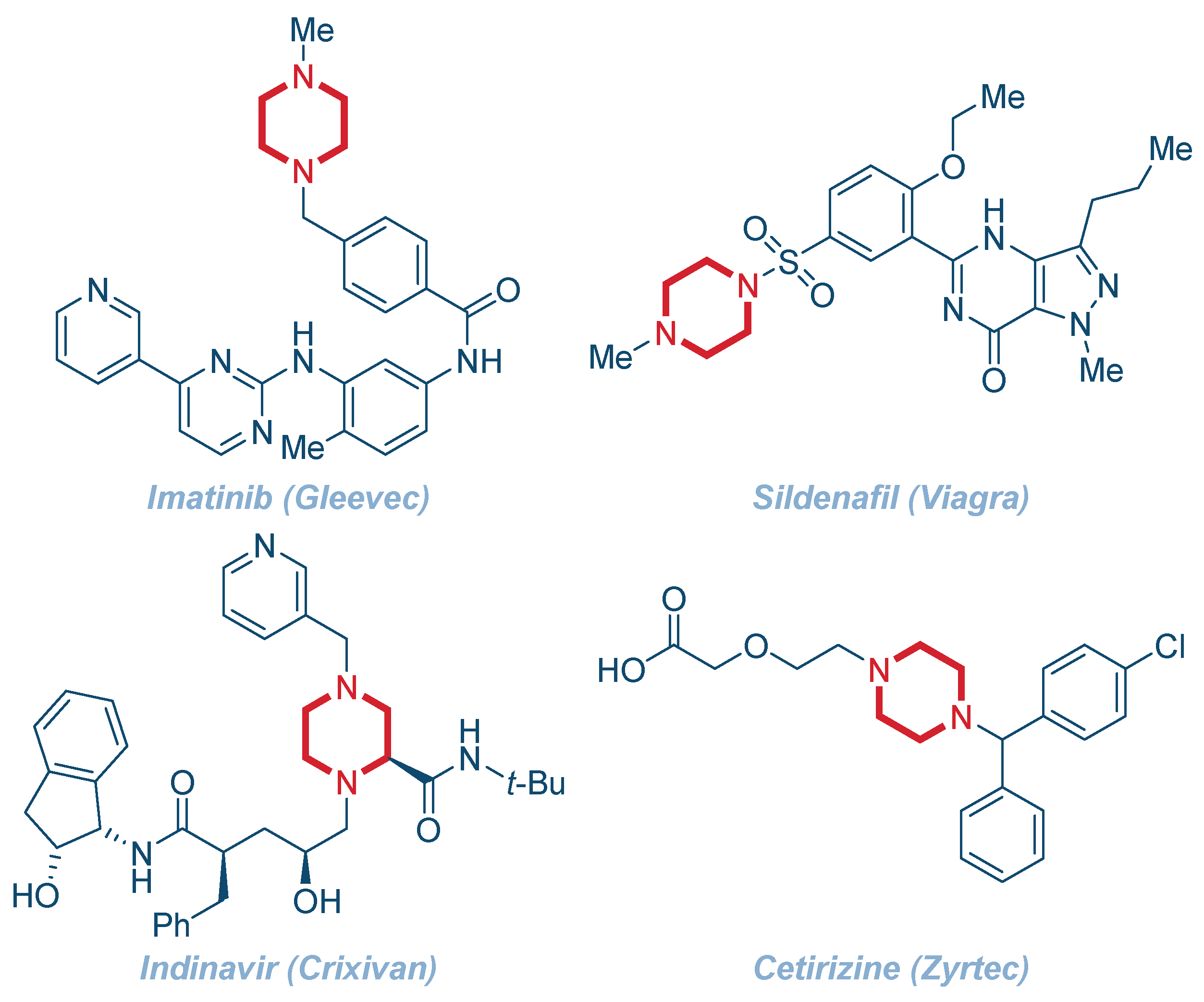
Organics | Free Full-Text | Recent Advances in the Synthesis of Piperazines: Focus on C–H Functionalization

Piperazine ring formation by a single-module NRPS and cleavage by an α-KG-dependent nonheme iron dioxygenase in brasiliamide biosynthesis | SpringerLink
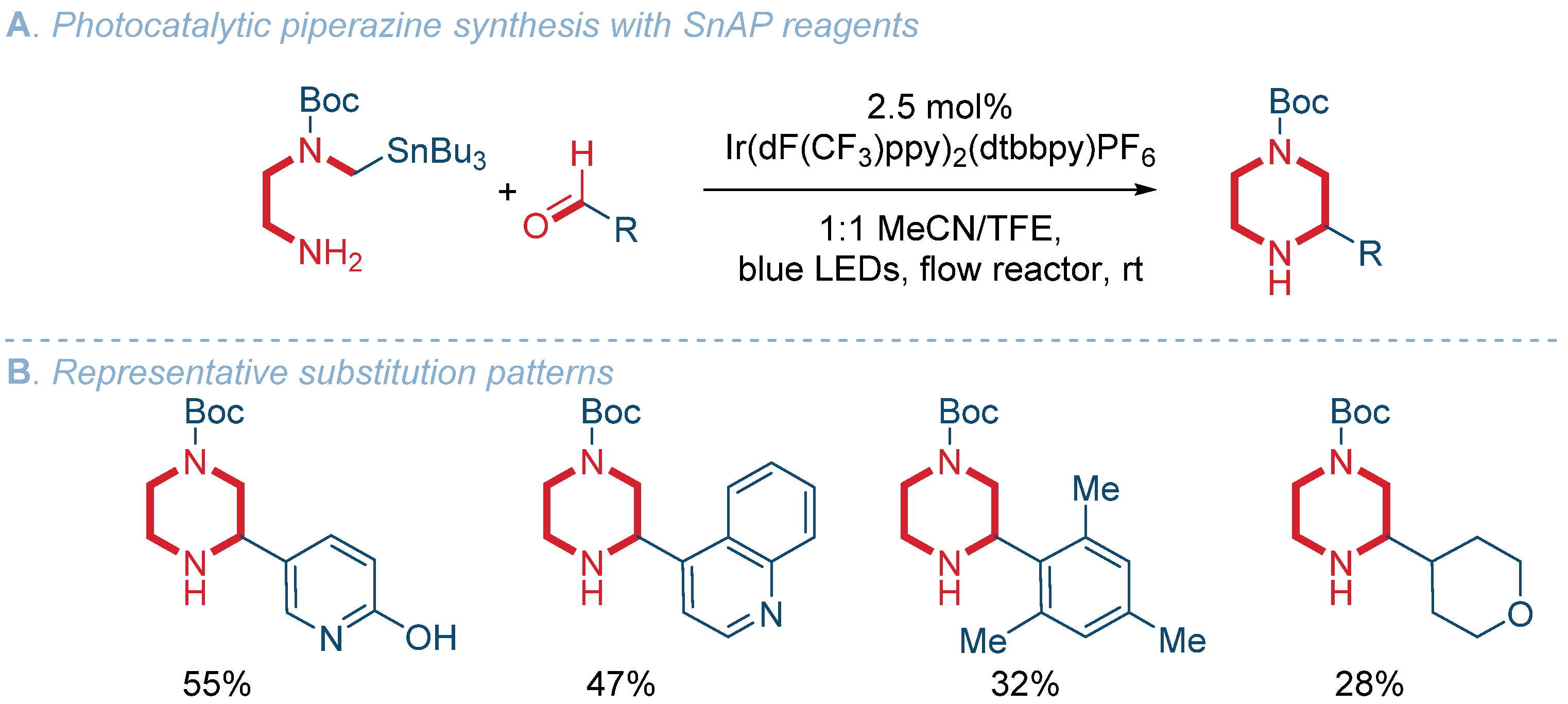
Organics | Free Full-Text | Recent Advances in the Synthesis of Piperazines: Focus on C–H Functionalization

Suggestion for N-substitution on the piperazine ring · Issue #8 · OpenSourceMycetoma/Series-1-Fenarimols · GitHub

Amazon | The 2016 World Market Forecasts for Imported Heterocyclic Compounds with Nitrogen Hetero-Atom(s) Only, Containing a Pyrimidine Ring, Piperazine Ring, or Unfused Triazine Ring, and Nucleic Acids and Salts | International,

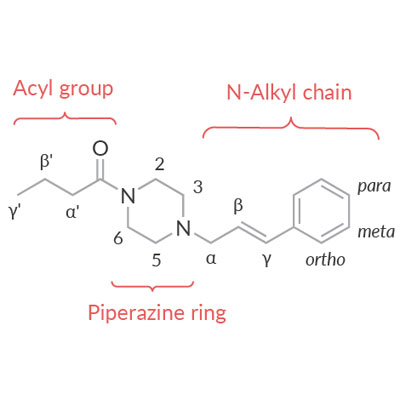


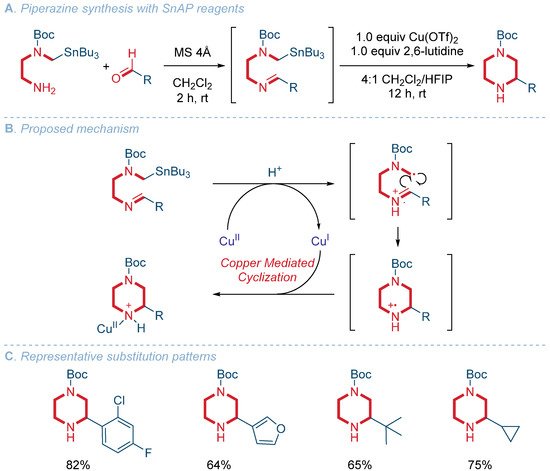



![Piperazine Nucleus Based Clinically Used Drugs [56-58]. | Download Table Piperazine Nucleus Based Clinically Used Drugs [56-58]. | Download Table](https://www.researchgate.net/profile/Anil-Kumar-Sharma-2/publication/235963303/figure/tbl1/AS:671532217020435@1537117165941/Piperazine-Nucleus-Based-Clinically-Used-Drugs-56-58_Q320.jpg)